Blog
Here to Strengthen Partnerships
We’re excited to welcome Craig to the CiiTA team—a seasoned leader whose diverse background and collaborative mindset align perfectly with our mission to empower pharmaceutical manufacturers with smarter, more actionable…
Read NowHidden 340B Duplicate Discounts
A national pharmaceutical vendor was facing a costly and complex issue—duplicate discounts on 340B inventory leading to millions in lost revenue. With non-Medicaid rebates mistakenly applied to 340B claims, the…
Read NowAn Extreme Outlier
CiiTA has a five-year partnership with a client to monitor and mitigate their 340B duplicate discount risk on an ongoing basis. During a recent evaluation of purchasing and Medicaid rebate…
Read NowState Processing Error
It is CiiTA’s firm belief 340B stakeholders are generally well-intentioned. However, it is very common for errors to occur within Medicaid claims processing, and manufacturers must maintain a constant vigilance…
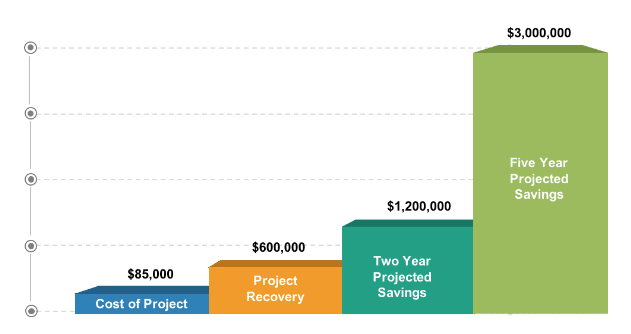
Read NowDuplicate Discount Project
CiiTA is frequently asked to develop and implement proof of concept pilot projects for clients who are just beginning to formalize their 340B management plans. In this case study, the…
Read Now